
The 5-year PFS rates were 10.8% vs 3.5%, respectively. Scores ranging from 1% to 49% were present in 37.1% and 37.0% of patients, respectively.Īdditional findings from the long-term analysis showed that the estimated 5-year OS rates were 18.4% in the combination arm compared with 9.7% in the placebo arm. Patients had a PD-L1 TPS of less than 1% at a rate of 34.2% and 35.2%, respectively. Most patients in both arms had a PD-L1 tumor performance score (TPS) of at least 1% (63.3% vs 63.0%), including 26.3% and 26.0%, respectively, who had a score of 50% or greater. Most patients in both arms were men (79.1% vs 83.6%), were not from East Asia (80.6% vs 81.5%), had an ECOG performance status of 1 (73.7% vs 68.0%), and were former or current smokers (92.1% vs 93.2%). Time from randomization to subsequent disease progression following the next line of treatment or death (PFS2) was an exploratory end point.īaseline patient characteristics were well-balanced between the combination and placebo arms the median age was 65.0 years (range, 29-87) vs 65.0 years (range, 36-88), respectively. Key secondary end points included overall response rate (ORR), duration of response (DOR), and safety. OS and PFS represented coprimary end points. In the combination arm, 55 patients completed 35 cycles of pembrolizumab and 12 started a second course of treatment with the agent. In the placebo arm, 172 patients received subsequent anticancer therapy, including 117 who crossed over to pembrolizumab monotherapy on-study and 26 who received anti–PD-L1 therapy. In the combination arm, 109 patients went on to receive subsequent anticancer treatment, including 33 who were treated with anti–PD-L1 therapy and 12 who received on-study second-course pembrolizumab. Additionally, patients were eligible to receive second-course pembrolizumab monotherapy for 17 cycles in the event of progression following completion of 35 cycles of pembrolizumab if a best response of stable disease or better per investigator assessment was achieve after at least 8 cycles of pembrolizumab. 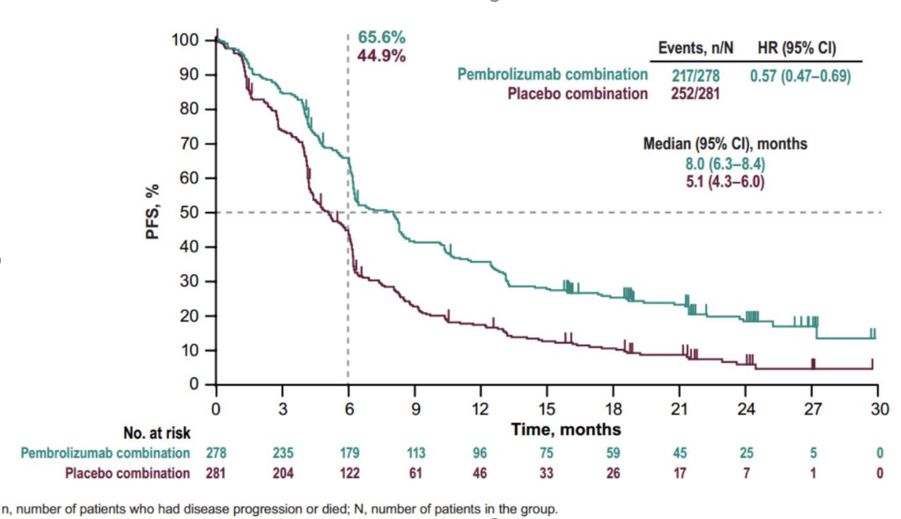
Patients in the placebo arm with disease progression confirmed by blinded independent central review were permitted to crossover to monotherapy with pembrolizumab for up to 35 cycles. 
Following completion of chemotherapy, patients received pembrolizumab 200 mg or placebo according to initially assigned treatment until the completion of 35 cycles, disease progression, unacceptable adverse effects (AEs), or withdrawal. Eligible patients were randomly assigned 1:1 to receive either pembrolizumab at a dose of 200 mg every 3 weeks in combination with carboplatin area under the curve 6 mg/mL/minute plus paclitaxel 200 mg/m2 or nab-paclitaxel 100 mg/m2 on days 1, 8, and 15 every 3 weeks for 4 cycles or placebo plus the same chemotherapy regimen. The multicenter, double-blind KEYNOTE-407 trial enrolled patients with stage IV squamous NSCLC who had measurable disease per RECIST 1.1 criteria and provided tumor tissue for investigators to determine PD-L1 status. Previously, results from KEYNOTE-407 led to the FDA approval of pembrolizumab in combination with carboplatin and paclitaxel or nab-paclitaxel as a frontline treatment for patients with metastatic squamous NSCLC on October 30, 2018. 
The median time from randomization to data cutoff was 56.9 months (range, 49.9-66.2). 1įindings from the 5-year KEYNOTE-407 safety and efficacy analysis showed that patients in the intention-to-treat population who were treated with pembrolizumab plus chemotherapy (n = 278) experienced a median overall survival (OS) of 17.2 months (95% CI, 14.4-19.7) compared with 11.6 months (95% CI, 10.1-13.7) among the 281 patients who received placebo plus chemotherapy (HR, 0.71 95% CI, 0.59-0.85). Patients with treatment-naïve metastatic squamous non–small cell lung cancer (NSCLC) experienced a clinically meaningful survival benefit when treated with the PD-1–directed antibody pembrolizumab (Keytruda) in combination with chemotherapy compared with those who received chemotherapy alone, according to long-term follow-up data from the phase 3 KEYNOTE-407 trial (NCT02775435) published in the Journal of Clinical Oncology.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
